 We may think from the above sketch that we have found our perfect Lewis Structure but there is one step left. 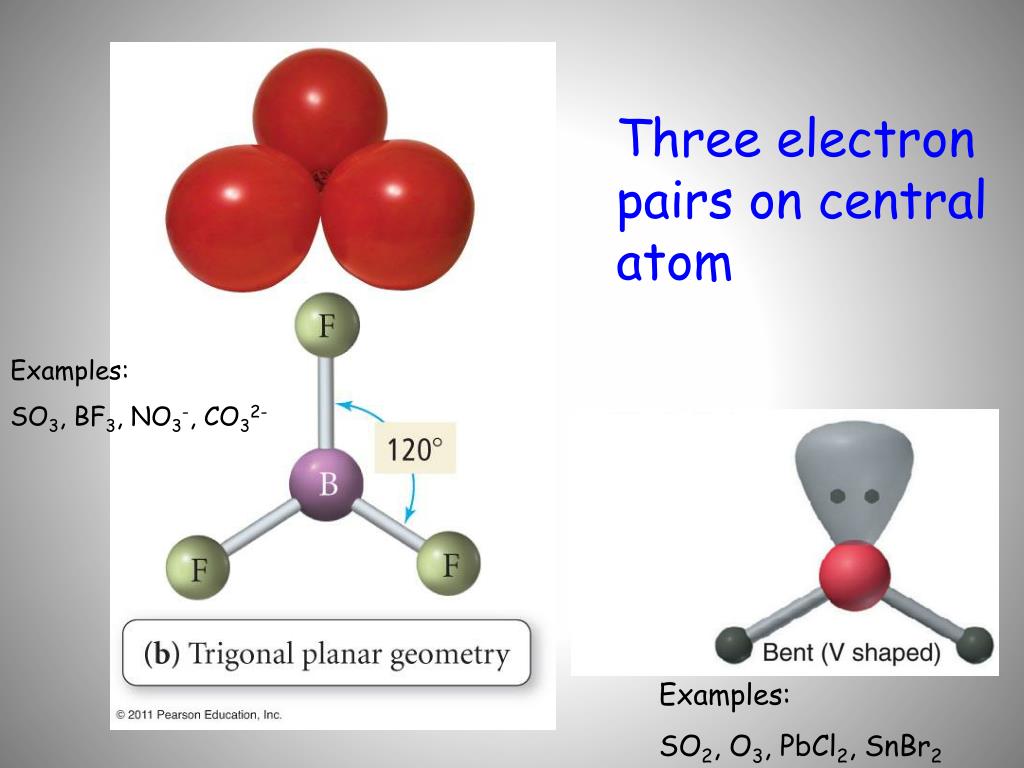
By drawing single bonds, we get the structure as: So, when atoms of different types come to combine in a molecular form, they follow the octet rule.Īs we see in the above-mentioned diagram, the five molecules have fulfilled their octet configuration and the total valence electron number remains 32. Here, we have put the symbols of sulfur and oxygen as per notations and put the valence electrons as dots.Ītoms present in the main groups of the periodic table tend to have a valency of eight following noble gases like argon, xenon, and so on. We are now going to sketch the skeletal diagram of sulfate ion with the help of atomic symbols and dot line structures. Step 3: Draw the skeleton diagram of the molecule Therefore, S or sulfur is the central atom. Here, between sulfur and oxygen, oxygen is more electronegative. To find out the central atom of the molecule, we have to consider electronegativity.Įlectronegativity is the measurement of an atom’s tendency to attract negatively charged electrons to form anionic molecules. Note: We had to add two electrons due to the negative 2- charge on sulfate ion. Total valence electrons in SO42- = 6*1 + 6*4 +2 = 32 Sulfur and oxygen both belong to the same group in the periodic table( the chalcogen family) and have six valence electrons each. Step 1: Count the total number of valence electrons present in the molecule/ionįor sulfate ions, we have one molecule of sulfur and four molecules of oxygen. Here, we will work with sulfate ion i.e SO42. The Lewis Structure is therefore also known as electron dot structure and is one of the most predominant and simplest concepts to understand the chemical bonding of molecular compounds. valence shell electrons to be dots and the bonds formed between atoms as straight lines. When we discuss chemical bonding through the Lewis Structure concept, we consider the electrons of the outermost shell i.e. The electrons, which are negatively charged particles, are present in shells surrounding the atomic nuclei. 
We must learn about the chemical bonding and additional features ofSO42- so that we have a clearer image and idea about nature and atomic reactions.Īs per the internal structure of a molecule, we know that a molecule is composed of atoms which in turn is composed of a nucleus and electrons. Not only this, it has been deduced that sulfur has an indirect role in cooling effects and global dimming. It plays an important factor in acid rain composition. Since we can easily get hold of this ion, be it naturally or synthetically, this helps us in our daily lives in a lot more ways than you can think of right now!įrom body and hygiene-care products like toothpaste, shampoos, soaps, and detergents to water treatment procedures, we can find the application of sulfate compounds everywhere.Īlthough it can be used in several facilities, sulfates have their drawbacks as well. Zn + H2SO4 -> H2 + ZnSO4 ( Here we get zinc sulfate by treating zinc metal with sulfuric acid.) We can also use sulfuric acid and metals to get our desired sulfate salts. We can easily prepare sulfates via oxidizing metal sulfites and sulfides. We find sulfates in a wide range of compounds, some of the well-known being MgSO4, CaSO4, Na2SO4, and PbSO4. 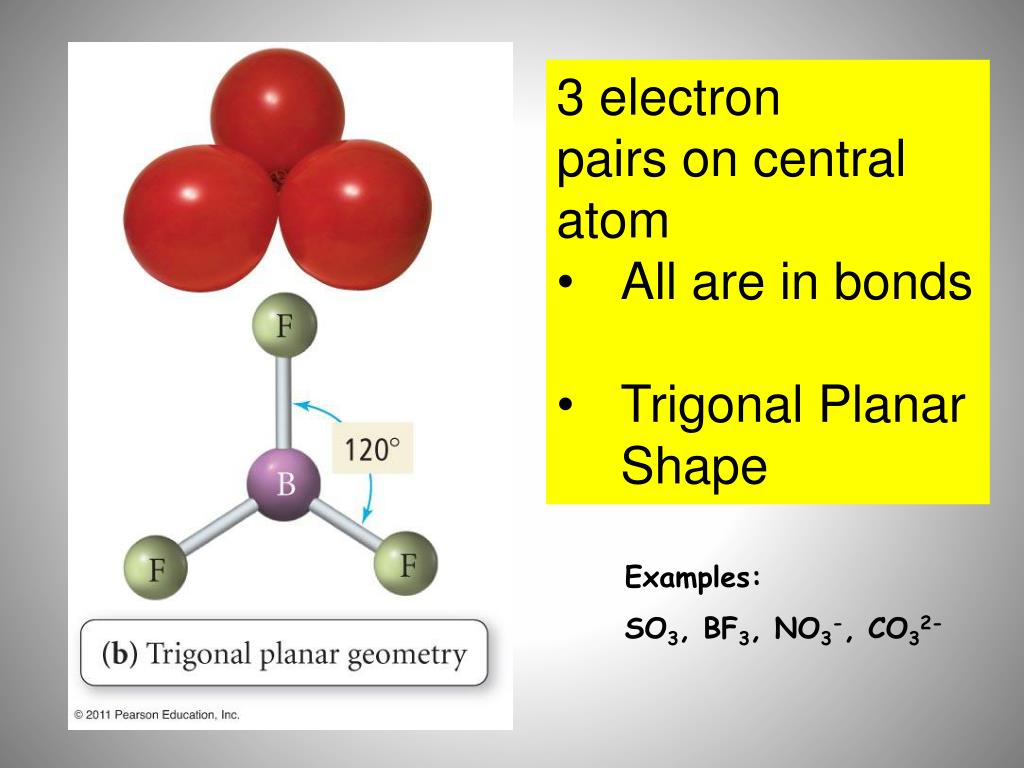
This is a polyatomic anion having a negative charge of -2. 1976.Sulfate ion (SO42-) is one of the most common ions that people in chemistry need to deal with. Landolt-Bornstein: Group II: Atomic and Molecular Physics Volume 7: Structure Data of Free Polyatomic Molecules. "Microwave Spectrum of Methyl Nitrate." Journal of Chemical Physics. Please address comments about this page to W. NIST does not necessarily endorse the views expressed, or concur with the facts presented on these sites.įurther, NIST does not endorse any commercial products that may be mentioned on these sites. There may be other web sites that are more appropriate for your purpose. No inferences should be drawn on account of other sites being referenced, or not, from this page. We have provided these links to other web sites because they may have information that would be of interest to you. Internal coordinates (distances (r) in Å) (angles (a) in degrees) (dihedrals (d) in degrees)Įxamples: C-C single bond, C=C, double bond, C#C triple bond, C:C aromatic bondīy selecting the following links, you may be leaving NIST webspace. See section I.F.4 to change rotational constant units A Listing of experimental geometry data for CH 3NO 3 (Methyl nitrate) Rotational Constants (cm -1) 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
